 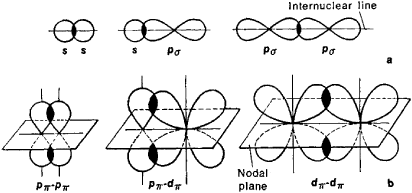
In the case of nitrogen in ammonia, this means hybridizing to have four equivalent sp3 orbitals, three of which are used for bonding with hydrogen atoms, and one of which holds the lone pair of electrons. * Energy*: In order to have the most stable (lowest energy) configuration, atoms will hybridize their orbitals to allow for maximum bonding. Hybrid orbitals (sp3 in this case) can form stronger, more stable overlapping (and thus bonds) than the pure s and p orbitals. * Overlapping*: When it comes to forming covalent bonds, the efficiency of orbital overlap is crucial. However, when nitrogen uses sp3 hybrid orbitals to bond, it can achieve the tetrahedral electron-pair geometry (with bond angles about 109.5 degrees) that matches the observed geometry. If nitrogen used its three 2p orbitals as they are, we would expect the bonds to be 120 degrees apart, resulting in a planar molecule, which is not the case. * Geometry*: The geometry of an ammonia molecule (NH3) is trigonal pyramidal, not trigonal planar. Instead, it "hybridizes" them with its 2s orbital to form four "sp3" hybrid orbitals.  
When a nitrogen atom forms an ammonia molecule, it doesn't use its three half-filled 2p orbitals separately to form bonds. This is a great question that goes into the details of molecular orbital theory and chemical bonding. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
